TULSA Procedure: Transurethral Ultrasound Ablation (TULSA-PRO) for Prostate Cancer
Home » About Prostate Cancer » TULSA® for Prostate Cancer
What is TULSA Treatment for Prostate Cancer?
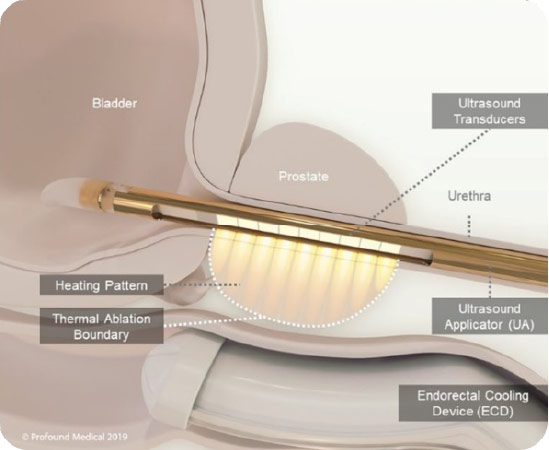
Transurethral ultrasound ablation (TULSA-PRO) is a minimally invasive procedure that, under MRI guidance, delivers precise doses of therapeutic ultrasound to treat prostate cancer while sparing the healthy nerve tissue surrounding the prostate. The TULSA-PRO procedure relies on a rod-shaped device that is inserted into the urethra.
TULSA Procedure vs. Other Prostate Cancer Treatments
Transurethral ultrasound ablation, or TULSA-PRO, can more easily cover a wider area of the prostate than laser focal therapy (LFT) or focal laser ablation (FLA). For patients who have a larger cancer, or a somewhat diffuse cancer within the prostate, the TULSA prostate procedure can cover almost the entire prostate gland in many patients.
Real-Time
Imaging
Directional Thermal
Ultrasound
Closed-Loop Temperature
Feedback
How does TULSA Procedure for Prostate Cancer Treatment Work?
Prostate Laser Center (a HALO Diagnostics Company) performs this procedure by combining real-time 3T MRI with computer control and robotically-driven directional thermal ultrasound energy. By doing so, this procedure can ablate (or destroy) cancerous tissues in the prostate. As a cancer treatment, TULSA doesn’t use incisions or radiation.
The TULSA procedure for prostate cancer allows the doctor to carefully and effectively target prostate tissue, tailoring the ablation to each patient. Once the cancerous areas in the prostate are identified and mapped, a computer controls the ablation with real-time temperature feedback.
Cancer Treatment: TULSA Procedure & Beyond
After the placement of the device in the prostate and the initial MRI scanning, the physician can begin a treatment plan. The TULSA-PRO system then individually controls ten separate elements, allowing for broad tissue destruction while fine-tuning each treatment area. This is all performed under the watchful eye of the physician, who can make adjustments as appropriate.
Advantages of TULSA-PRO
Decreased Risk of Side Effects
TULSA-PRO has a decreased risk of erectile and urinary side effects compared to whole gland therapy.[1]Same-Day Outpatient Procedure
TULSA-PRO is performed in a single session that takes a few hours.Keep Your Options Open
If you are in need of future prostate care, you may either have a repeat TULSA procedure or choose any other type of prostate therapy to address your prostate needs.[2]Advanced Image Guidance
Directional thermal ultrasound and MR imaging with active heat monitoring.Customizable Treatment Plans
TULSA-PRO is customizable and personalized based on your situation and goals.Shared Decision Making
Before your TULSA procedure, you will decide with your physician on a treatment plan.How Dr. Karamanian Performs the TULSA Procedure
The TULSA procedure (TULSA-PRO) ablates prostate tissue from the inside out. A device enters the urethra, which delivers thermal ultrasound energy outwards from the urethra to the prostate while under real-time MRI guidance.
This approach avoids the need for surgical incisions to reach the prostate. A cooling mechanism helps protect the urethra from the ultrasound energy. The thermal ultrasound energy is delivered in a sweeping motion (visualize a clock hand sweeping), which enables the physician to ablate a wider area.

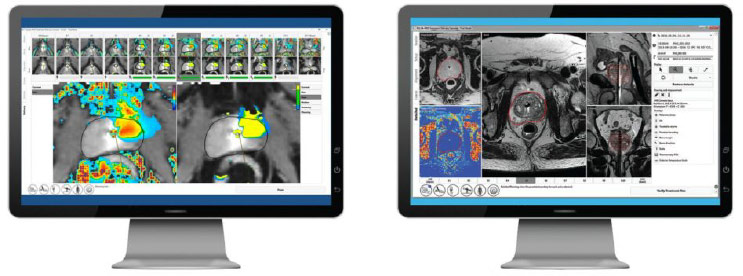
TULSA-PRO produces real-time temperature maps, enabling the physician to see the increasing temperature of the prostate tissue and surrounding structures every 5-7 seconds during treatment. This visibility allows the physician to actively monitor tissue heating.
What are the common side effects of the TULSA procedure?
The TULSA procedure is a minimally invasive surgery and is typically conducted as a same-day out-patient procedure. It has lower incidences of certain post-operative side effects as compared to some traditional prostate cancer treatments, such as radical prostatectomy (removal of the entire prostate gland) and radiation therapy.
However, as with any other treatment, procedure, or surgery, there are certain risks.
The most common side effects of the TULSA procedure include: pain/discomfort in the ablation area, blood in the urine, urinary tract infection, urinary incontinence, erectile dysfunction, and residual or recurrent prostate cancer. If you experience any of these side effects after undergoing the TULSA procedure, contact your doctor.
Frequently Asked Questions about the Tulsa Procedure
Can the TULSA procedure (TULSA-PRO) perform partial and whole gland ablation?
Our physicians can customize the procedure to ablate a specific area of the prostate or almost the entire prostate, depending on your needs and functional outcome goals. Customization allows for more targeted treatment for each individual patient.
Do I need to remain in a hospital or medical facility overnight after the TULSA procedure?
The TULSA procedure does not require an overnight stay following treatment. Patients can return to their hotel on the same day they are treated.
What are the side effects?
As with many procedures, there are side effects associated with the TULSA procedure. The most common side effects include pain/discomfort in the procedure area, blood in urine, urinary tract infection, urinary incontinence, and erectile dysfunction.
According to the TACT: TULSA® Ablation Clinical Trial conducted by Profound Medical (the makers of TULSA), the study found patients had a 2.6% urinary incontinence risk and 25% erectile dysfunction risk.
Who qualifies for TULSA-PRO?
Whether a patient qualifies for the TULSA procedure will be determined by the treating physician on a case-by-case basis. In general, patients who are good candidates for the TULSA procedure have low- to intermediate-risk disease that is confined to the prostate.
Generally, the patient should have a PSA of less than 20. If a patient has significant calcifications within the prostate (as determined by a CT scan), then they are not a candidate for TULSA, because the calcifications will block sound waves. Patients should be relatively healthy and able to undergo general anesthesia. Contact us if you would like more information.
What to Expect
TULSA for Prostate Cancer
Wondering what to expect before, during, and after your TULSA for prostate cancer procedure? Click here for more information.
References
[1] Hatiboglu G, Popeneciu V, Bonekamp D, Burtnyk M, Staruch R, Distler F, Radtke JP, Motsch J, Schlemmer HP, Pahernik S and Nyarangi-Dix J (2021) Single-Center Evaluation of Treatment Success Using Two Different Protocols for MRI–Guided Transurethral Ultrasound Ablation of Localized Prostate Cancer. Front. Oncol. 11:782546. doi: 10.3389/fonc.2021.782546
[2] Nair, S.M., Stern, N., Dewar, M. et al. (2020). Salvage open radical prostatectomy for recurrent prostate cancer following MRI-guided transurethral ultrasound ablation (TULSA) of the prostate: Feasibility and efficacy. Scandinavian Journal of Urology, 54(3), 215-219. doi: 10.1080/21681805.2020.1752795
Request a consultation today
If you’re curious about the TULSA procedure for prostate cancer and would like more information, request a consultation with the Prostate Laser Center today.

Interested in our treatment options?
Request a Consultation
